 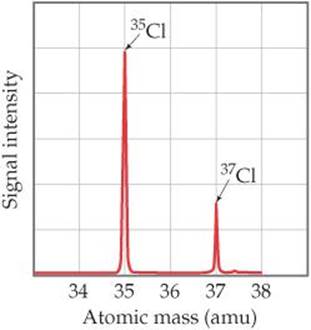
Isotope separation to produce pure 37 Cl can vastly reduce 36 Cl production, but a small amount might still be produced by (n,2n) reactions involving fast neutrons. There are two principal stable isotopes, 35 Cl (75.77) and 37 Cl (24.23), found in the relative proportions of 3:1 respectively, giving chlorine atoms in bulk an apparent atomic weight of 35.5. This produces a long lived radioactive product which has to be stored or disposed off. Chlorine ( Cl) has isotopes with mass numbers ranging from 32 g mol 1 to 40 g mol 1. In chloride-based molten salt reactors the production of 36 Cl by neutron capture is an inevitable consequence of using natural isotope mixtures of chlorine (i.e. 36Cl has seen use in other areas of the geological sciences, forecasts, and elements. The ratio of 36 Cl to stable Cl in the environment is about 700 x 10 -15 : 1 (Bentley et. Only three of these isotopes occur naturally: stable 35 Cl (75.77)and 37 Cl (24.23), and radioactive 36 Cl. Thus, as an event marker of 1950s water in soil and ground water, 36Cl is also useful for dating waters less than 50 years before the present. Chlorine has 9 isotopes with mass numbers ranging from 32 to 40. The residence time of 36Cl in the atmosphere is about 1 week. Additionally, large amounts of 36Cl were produced by neutron irradiation of seawater during atmospheric detonations of nuclear weapons between 19. The half-life of this hydrophilic nonreactive isotope makes it suitable for geologic dating in the range of 60,000 to 1 million years. 36Cl decays to either 36 S (1.9%) or to 36 Ar (98.1%), with a combined half-life of 308,000 years. In the subsurface environment, 36Cl is generated primarily as a result of neutron capture by 35Cl or muon capture by 40 Ca. 36Cl is produced in the atmosphere by spallation of 36 Ar by interactions with cosmic ray protons. Trace amounts of radioactive 36Cl exist in the environment, in a ratio of about 7×10 −13 to 1 with stable isotopes. The shortest-lived are proton-unbound 29Cl and 30Cl, with half-lives less than 10 picoseconds and 30 nanoseconds, respectively the half-life of 28Cl is unknown. All other isotopes have half-lives under 1 hour, many less than one second. The longest-lived radioactive isotope is 36Cl, which has a half-life of 301,000 years. Chlorine (Cl) has isotopes with mass numbers ranging from 32 g mol 1 to 40 g mol 1. There are two stable isotopes, 35Cl (75.77%) and 37Cl (24.23%), giving chlorine a standard atomic weight of 35.45. Chlorine stable isotope analysis was carried out on methyl chloride (CH3Cl) after converting AgCl into CH3Cl by reacting it with methyl iodide (CH3I). Find the average atomic mass of Chlorine. Chlorine-35 is three times more common than Chlorine-37 so the ratio is 3:1. Chlorine ( 17Cl) has 25 isotopes, ranging from 28Cl to 52Cl, and two isomers, 34mCl and 38mCl. Chlorine has two common stable Isotopes Chlorine-35 and Chlorine-37. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
